 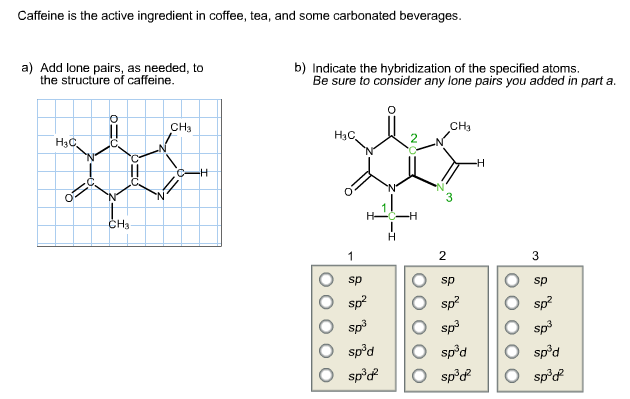 
Coffee, black and green tea, cocoa, cola soft drinks, and energy drinks contain caffeine.Ĭhocolate bars, energy bars, and some over-the-counter medications like cough syrup and slimming tablets are all available. White white What is the caffeine content of the beverage?Ĭaffeine is a natural substance found in the leaves and fruits of some plants. Is caffeine a prescription drug?Ĭaffeine provides a temporary energy boost to most people and improves their mood. Caffeine, as is known, has a stimulating effect on plants like coffee and tea. When pure, it’s an odorless white solid that can be partially soluble in water at temperatures between 234 and 236 degrees Celsius (100 mM). What kind of substance is caffeine in?Ĭaffeine is a xanthine alkaloid. Despite the fact that it is an aromatic compound, it still appears to be a ketone (organic compound with a carbonyl group) rather than an enol (alkene with a hydroxyl group), which is why it does not appear to be aromatic when viewed from its normal structure. Who Discovered Caffeine?įriedlieb Ferdinand Runge Friedlieb Ferdinand Runge Is caffeine a stimulant?Ĭaffeine is an alkaloid that is found in the body. It will raise water’s pH to some extent.īecause it is a weak base, the free base will be in equilibrium with its protonated counterpart. Why is caffeine used as a base?Ĭaffeine isn’t acidic. (4 pts) Caffeine is found in an average cup of coffee (Molecular formula: C8H10N4O2). Does 3 pairs beat a full house? What amount of caffeine does it contain?Ģ8.What is the advantage of twisted pair cable?.Why did mitchel musso leave pair of kings.In caffeine, how many valence electrons do you have?Ĩ valence electrons Eight valence electrons In caffeine, how many atoms do you have?Ĩ atoms of carbon, 10 atoms of hydrogen, 2 atoms of oxygen, and 4 atoms of nitrogen are found in one molecule of caffeine. They are primarily zwitterionic resonance structures in which nitrogen and carbon atoms are double-bonded to each other, making both nitrogen and carbon atoms essentially planar (in sp2 orbital hybridization). 
Is caffeine a plane molecule? Caffeine is a non-stereoisomeric achiral molecule.Ĭaffeine has two amide groups. Despite the fact that some are frequently drawn with three single bonds, these atoms’ lone pairs are involved in resonance with adjacent double-bonded carbon atoms and adopt an sp2 orbital hybridization as a result. The caffeine molecule’s nitrogen atoms are all planar. So, what exactly is caffeine hybridization? As you can see from the structure, the Oxygen and Nitrogen molecules have a higher polarity than Carbon, which allows them to pull electrons in their covalent bonds slightly. Is caffeine, on the other hand, a polar or nonpolar compound? Caffeine is a polar molecule, to be sure. After all, how many electrons does caffeine contain? Caffeine is aromatic because it has 10 electrons. Two lone pairs will be found in oxygen with two bonds and a full octet, while one lone pair will be found in nitrogen with three bonds and a full octet.Īs a result, caffeine is found in eight lone pairs. When smoked, cocaine reaches the brain in 15 s.Nitrogen and oxygen are the elements of interest in caffeine for single pairs uncharged carbons will not have lone pairs. It vaporizes at the temperature of a burning cigarette. Crack cocaine is more volatile than cocaine hydrochloride. \nonumber \]īecause it is soluble in water, cocaine hydrochloride is readily absorbed through the watery mucous membranes of the nose when it is snorted. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
